Case Report 
 Creative Commons, CC-BY
Creative Commons, CC-BY
A Case of Hypercalcemia Induced PRES in an Immobilized Adult
*Corresponding author: Brian Behnke, Department of Internal Medicine, HCA Health One Sky Ridge Medical Center, USA.
Received: May 17, 2022; Published: May 20, 2022
DOI: 10.34297/AJBSR.2022.16.002216
Introduction
Here we highlight the importance of serum calcium levels in a patient with prolonged immobility presenting with altered mental status. To our knowledge, there are no other published case reports of hypercalcemia induced posterior reversible encephalopathy syndrome (PRES) in the setting of prolonged immobility.
Case Report
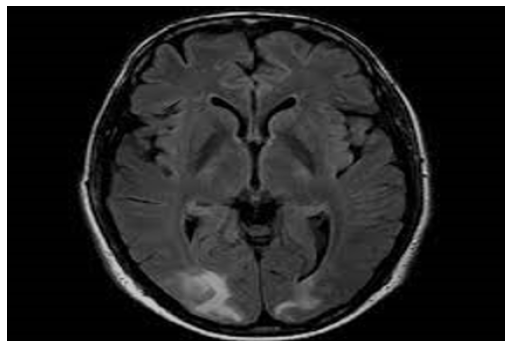
A 46-year-old male presented to an outside facility with depressed mental status, and hemoglobin of 3.1g/dL. The patient had a history of chronic flaccid paralysis secondary to Miller-Fisher variant GBS with tracheostomy and gastric tube dependence, seizures, deep venous thrombosis, and remote history of injection drug use. Other notable labs on initial presentation included: serum sodium of 160mmol/L, creatinine of 1.49mg/dL, calcium of 12.1mg/dL, and white blood cell count (WBC) of 17.1*109/L. The patient was normotensive to hypotensive throughout his stay in the outside facility and chest x-ray showed extensive bilateral patchy infiltrates. Two weeks prior to presentation, a computed tomography angiogram (CTA) of the chest showed diffuse consolidative changes throughout the lungs, a small pleural effusion, no pulmonary embolism. Per the outside facility report, and as was later corroborated with family, the patient was alert and oriented x3 at baseline and paralyzed from below the neck. Prior to transfer, the patient was transfused two units of packed red blood cells with relative stabilization of his anemia and mechanically ventilated via tracheostomy due to acute hypoxic respiratory failure. Brain imaging, including CTs and MRIs, showed confluent parietooccipital white matter hypodensity compatible with posterior reversible encephalopathy syndrome (Figure 1) [1-4]. Neurology was consulted upon arrival to our facility. Throughout the patient’s admission, his total serum calcium was persistently elevated and ionized calcium ranged 1.49 to 1.75mmol/L. Treatment with intravenous (IV) fluids, bisphosphonates and denosumab were required to correct patient’s hypercalcemia into the high normal range, but there was no noted change in mental status. During the patient’s hospital course, his renal function improved with intravenous fluid resuscitation. Given that the patient was normotensive to hypotensive, acute kidney injury resolved with fluids, and cytotoxic agents were excluded as potential triggers for PRES, it was determined that the patient’s persistent hypercalcemia was the most likely cause of his PRES syndrome. Despite aggressive medical interventions, the patient’s mental status did not improve and per family discussions, the patient was admitted to inpatient hospice for comfort measures only.
Discussion
We report a case of PRES in the setting of severe, refractory hypercalcemia of a critically ill patient. The pathophysiology relating hypercalcemia to PRES is that hypercalcemia causes cerebral vasoconstriction and endothelial dysfunction impairing cerebral autoregulation. Specifically, hypercalcemia has been shown to directly alter contractile tone of the endothelium and underlying smooth muscle cells leading to increased vascular resistance. Additionally, both animal models and human experiments have shown that hypercalcemia induces an inflammatory response in endothelial and leukocytes, which leads to endothelial dysfunction. An additional point of discussion in this case is that our patient lacked typical causes of hypercalcemia. Our patient had no evidence of hyperparathyroidism syndromes [5], vitamin D abnormalities, nor medications causing hypercalcemia, thus immobility was suspected as the most likely cause of outpatient’s hypercalcemia. There have been a few case reports published that demonstrate the association between hypercalcemia and immobility, especially in young adult males with peak bone mass following sudden neurological injuries. Spinal cord injury has been noted in multiple studies to be associated with rapid and extensive bone loss, especially distal to the spinal cord lesion [6]. Certain specific osseous sites have been found to have bone loss at approximately 1% per week for the first year following a spinal cord injury.
It is thought that the association between prolonged immobility and hypercalcemia is likely multifactorial in etiology. One factor that has been hypothesized to affect this association is that the complete and sudden loss of mechanical forces on bone leads to increased osteoclastic activity. This has been seen in space exploration whereby it has been noted that astronauts lose 1 to 2% of their bone density every month due to the loss of gravity, which applies a constant mechanical force to the skeletal system. Without any mechanical forces applied, bones are unable to remodel themselves to maintain certain density to support the body [7- 11]. Another contributing factor of prolonged immobilization with hypercalcemia is thought to be due to the increased production of sclerostin from osteocytes, which suppresses the Wnt-Runx2 pathway, leading to decreased osteoblastic bone formation. In fact, recent studies done on rat models have shown that administration of sclerostin-neutralizing antibodies as quickly as 7 days after a spinal cord injury and continued for as many as 7 weeks has been shown to dramatically increase bone formation and bone mass in skeleton distal to the spinal cord lesion.
Conclusion
Although there are multiple etiologies for a patient who presents with PRES, our case emphasizes the importance of not overlooking the value of the serum calcium level, especially in the setting of a young male adult with prolonged immobility.
References
- Wilson MP, Jack AS (2020) Coronavirus disease 2019 (COVID-19) in neurology and neurosurgery: A scoping review of the early literature. Clin Neurol Neurosurg 193: 105866.
- Almufarrij I, Uus K, Munro KJ (2020) Does coronavirus affect the audio-vestibular system? A rapid systematic review. Int J Audiol 59(7): 487-491.
- Brouwer MC, Ascione T, Pagliano P (2020) Neurologic aspects of covid-19: a concise review. Infez Med 28(Suppl 1): 42-45.
- Malayala SV, Gisha Mohan, Deepa Vasireddy, Paavani Atluri (2021) A case series of vestibular symptoms in positive or suspected COVID-19 patients. Infez Med 29(1): 117-122.
- Mao L, Huijuan Jin, Mengdie Wang, Yu Hu, Shengcai Chen, et al. (2020) Neurological manifestations of hospitalized patients with COVID-19 in Wuhan, China. JAMA Neurol 77(6): 683-690.
- Arabi Y, Harthi A, Hussein J, A Bouchama, Johani S, et al. (2015) Severe neurologic syndrome associated with Middle East respiratory syndrome corona virus (MERS-CoV). Infection 43(4): 495-501.
- Saniasiaya J, J Kulasegarah (2021) Dizziness and COVID-19. Ear Nose Throat J 100(1): 29-30.
- Bertholon P, Thai Van H, Bouccara D, Esteve Fraysse MJ, Wiener Vacher SR, et al. (2021) Guidelines of the French Society of Otorhinolaryngology (SFORL) for teleconsultation in patients with vertigo during the COVID-19 pandemic. Eur Ann Otorhinolaryngol Head Neck Dis 138(6): 459-465.
- Lee H, SI Sohn, YW Cho, SR Lee, BH Ahn, et al. (2006) Cerebellar infarction presenting isolated vertigo: frequency and vascular topographical patterns. Neurology 67(7): 1178-1183.
- Kattah JC, Arun V Talkad, David Z Wang, Yu Hsiang Hsieh, David E Newman Toker, et al. (2009) HINTS to diagnose stroke in the acute vestibular syndrome: three-step bedside oculomotor examination more sensitive than early MRI diffusion-weighted imaging. Stroke 40(11): 3504-3510.
- Hegemann SC, A Wenzel (2017) Diagnosis and treatment of vestibular neuritis/neuronitis or peripheral vestibulopathy (PVP)? Open questions and possible answers. Otol Neurotol 38(5): 626-631.
- Dessau R, G Lisby, J Frederiksen (1999) Coronaviruses in spinal fluid of patients with acute monosymptomatic optic neuritis. Acta Neurol Scand 100(2): 88-91.
- Yeh EA, Arlene Collins, Michael E Cohen, Patricia K Duffner, Howard Faden, et al. (2004) Detection of coronavirus in the central nervous system of a child with acute disseminated encephalomyelitis. Pediatrics 113(1): e73-e76.
- Viola P, Massimo Ralli, Davide Pisani, Donatella Malanga, Domenico Sculco, et al. (2020) Tinnitus and equilibrium disorders in COVID-19 patients: preliminary results. Eur Arch Otorhinolaryngol 278(10): 3725-3730.



 We use cookies to ensure you get the best experience on our website.
We use cookies to ensure you get the best experience on our website.